
My reported number was around 3050 which fell in between the range of 1020-3100. Alkene – Xylene : Group A represents a alkene with an absorption intensity of medium. Not all of the named compoundsnecessarily matches up with a spectra. More than one (or none) of thesecompounds might be a possible match for a single spectra. 950 – N/A 5: Match the following with their possible spectra above (A – D). 1710 (RCOR) – Ketone – Strong OR Acid (RCOOH) - Strong 4 : List the major peaks in each IR spectrum and what functional groups could be (30pts) A: 
There are four IR spectra (see another file). Based on my numbers I believe IR spectrum of cyclohexanol (thin film) is similar to my results, although based on the looks of the graph human error may have changed me sample. Finally, I compare my results with the chart in the lab manual. We can also see that we have carbon and hydrogen bonds present because we have peaks between 2900cm-cm-1. A small peak was observed and it was approximately 3400cm-1, this shows us that there is a hydroxyl group present. On the infrared spectrum there were many peaks. 3: Observation: Once the unknown liquid was analyze a graph appeared and we had to compare it to the chart to identify our unknown liquid compound. Place this assembly into the spectrometer. Place the dicks into the liquid sample holder and replace top. Place the disks together, placing compound in between the disk like a sandwich. Apply half a drop to only one of the disks. 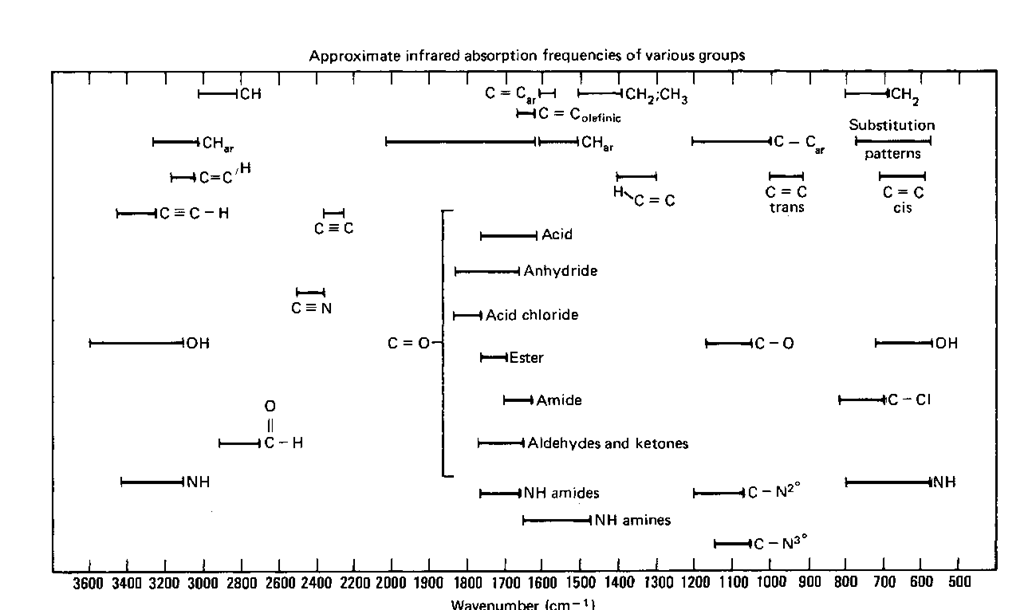
2: Procedure: Place NaCl disks flat on preparation tray. This procedure uses the vibration of atoms to measure the molecule to determine the functional group. Section 18935 Analysis of Infrared Spectra Name: Shahid Waseem Date:04-01- 1: Purpose (10pts) The purpose of this experiment is to identify an unknown liquid by using the infrared analysis of IR Spectra.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
